 
Hybridization of the 2 s and two of the 2 p AOs forms three sp 2 hybrid orbitals, oriented 120° with respect to each other in the same plane one of the 2 p AOs remain unhybridized (move the slider around to see the before/after of hybridization).Ĭombining one valence s AO and all three valence p AOs produces four degenerate sp 3 hybridized orbitals, as shown in Figure 4 for the case of 2 s and 2p AOs. Energetically, sp 2 hybrid orbitals lie closer to the p AO than the s AO, as illustrated in Figure 2 (the sp 2 hybrid orbitals are higher in energy than the sp hybrid orbitals).įigure 2. The sp 2 hybrid orbitals have twice as much “ p” character as “ s” character this is indicated by the superscript “2” in sp 2. After hybridization, there is one unhybridized 2 p AO left on the atom. Each hybrid orbital is pointed toward a different corner of an equilateral triangle. The three sp 2 hybrid orbitals are oriented at 120° with respect to each other and are in the same plane-a trigonal planar (or triangular planar) geometry. However, in a covalent molecule, the one large lobe of each sp hybrid orbital gives greater overlap with another orbital from another atom, yielding σ bonds that lower the molecule’s energy.Ĭombining one valence s AO and two valence p AOs produces three degenerate sp 2 hybrid orbitals, as shown in Figure 2 for the case of 2 s and 2p AOs. For example, a beryllium atom is lower in energy with its two valence electrons in the 2 s AO than if the electrons were in the two sp hybrid orbitals. The hybridized orbitals are not energetically favorable for an isolated atom. (Move the slider to see the before/after of hybridization). Specifically, the sp hybrid orbitals’ relative energies are about half-way between the 2 s and 2 p AOs, as illustrated in Figure 1.įigure 1 Hybridization of the 2 s and one of the 2 p AOs forms two sp hybrid orbitals, oriented 180° with respect to each other the two other 2 p AOs remain unhybridized. One of the ways in which the hybrid orbitals exhibit their mixed “ s” and “ p” characteristics is in their energy. After the hybridization, there are two unhybridized 2 p AOs left on the atom.īecause these hybrid orbitals are formed from one s AO and one p AO, they have a 1:1 ratio of “ s” and “ p” characteristics, hence the name “ sp“. 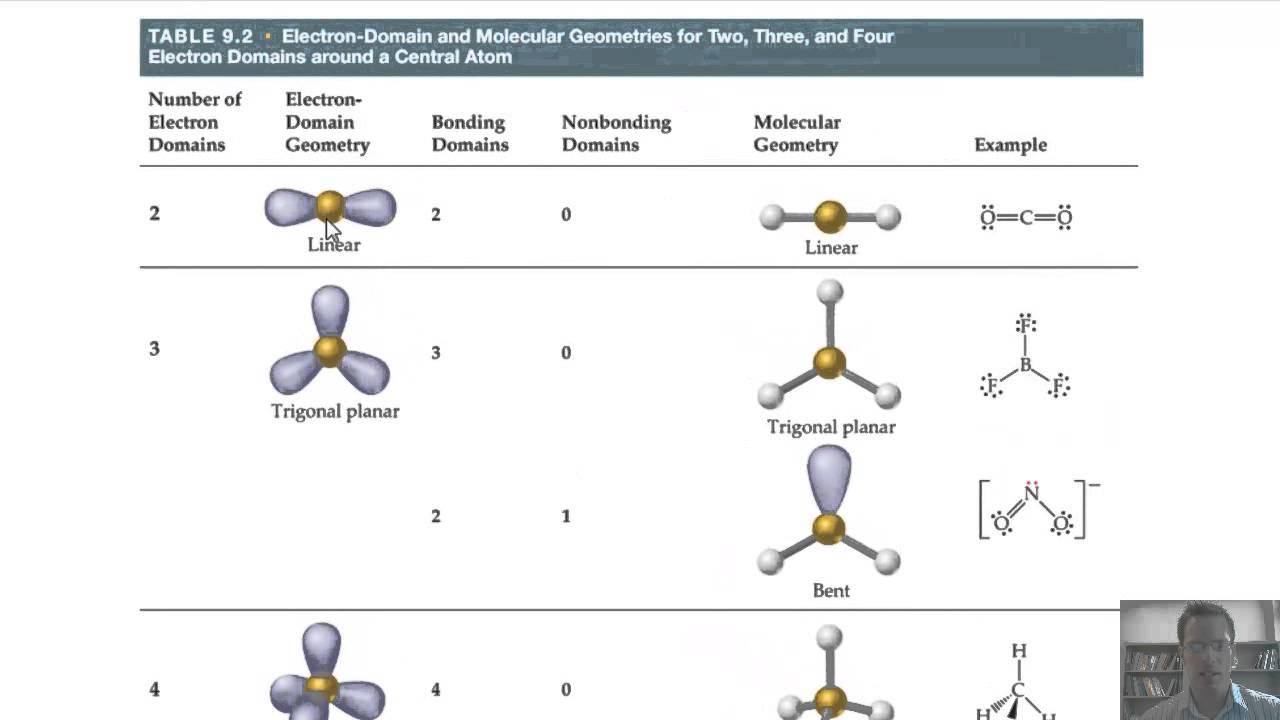
The two sp hybrid orbitals are oriented at 180° to each other-a linear geometry.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
